Basic Chemistry and Terminology
This section is likely the least accessible to general audiences – basic concepts in chemistry and geology may be explored through links in the text. Some of the concepts involved in specific analytical techniques need only be understood at the conceptual level.
Marble is composed of two minerals: calcite (calcium carbonate or CaCO3) and less commonly dolomite (calcium magnesium carbonate or CaMg(CO3)2). Because calcium and magnesium are both alkaline earth metals–they occupy the same column in the periodic table of elements and therefore have the same number of free electrons–magnesium replaces some of the calcium in dolomite.
Crystal structure of calcite and dolomite. Note: both minerals have many different possible configurations and shapes; these models illustrate the relationship between chemical and structural differences. (Images from mindat.org - see footnote [1])
Marble is chemically identical to limestone (both are made of "calcareous" stone i.e. calcium carbonate), but they are structurally different. Limestone is a sedimentary rock, meaning it was formed by the deposition of bony material on the Earth’s surface (often an ancient seabed). Marble is a metamorphic rock, meaning it was formed from sedimentary rock that was deep under the surface of the Earth, where pressure and heat compressed the crystal structure, before it was brought to up by geological activity like volcanism or plate tectonics.
Marble often contains impurities in the form of trace elements, especially iron (Fe), magnesium (Mg), manganese (Mn), Zinc (Zn), and strontium (Sr). Pure calcite and dolomite are white, and these impurities can create colored marbles.[1] Some specific quarry sites have unique features – the classic example is Proconnesian marble, which often contains sulfuric gases trapped in the rock that produce a rotten egg smell when the marble is broken.[2]
Almost all scientific studies of ancient marble have been undertaken by members of the Association for the Study of Marble and Other Stones in Antiquity (ASMOSIA), formed in 1988 during a conference organized by many of the scientists collaborating in the developing field.
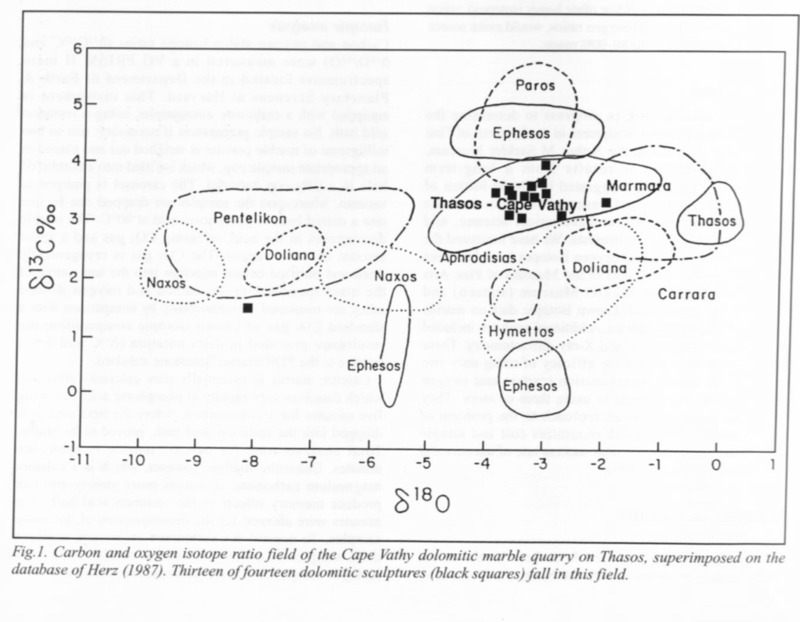
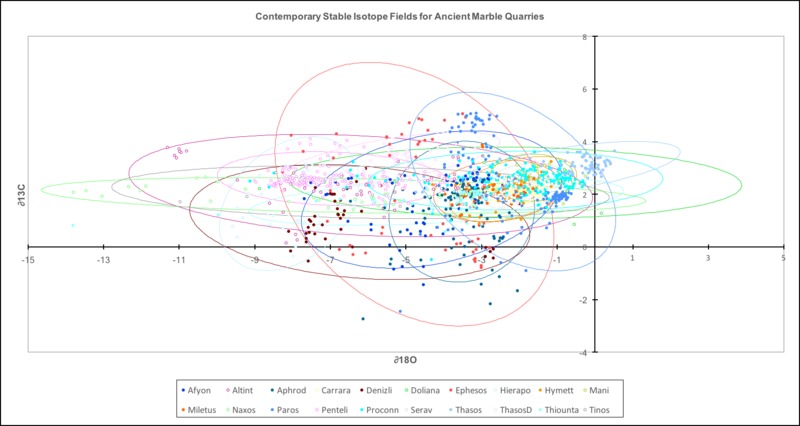
The most important technique in marble analysis has been stable isotope analysis, often attributed to Norman Herz’s and David Wenner’s early work in the field.[3] The method looks at the ratio of two isotopes of carbon and oxygen; Carbon-13 to Carbon-12 and Oxygen-18 to Oxygen-16. This technique showed promise because it was a simple, relatively cheap test that required only small samples of stone. Scientists took the ratios of the two isotopes (usually given as ∂13C and ∂18O – a notation expressing relative quantities for comparison) for many samples of marble. Stable isotope analysis uses a universal standard - the ratios of C13 to C12 and O18 to O16 are set to those in Peedee Belemnite (PDB) - a Cretaceous marine fossil found in the Peedee Formation in South Carolina. For example, the data point for the Young Hero or God (formerly known as Meleager object 1926.48) is +2.56 for ∂13C and -3.36 for ∂18O – this means that the ratio of Carbon-13 to Carbon-12 is 2.56 parts per thousand higher than in PDB, while the ratio of Oxygen-18 to Oxygen-16 is 3.36 parts per thousand less than in PDB. Today, PDB is no longer used, but several chemically identical standards have been created such as Vienna Peedee Belemnite and NBS-19 limestone.
From this data, graphs could be drawn that showed different ranges of stable isotope ratios for marble - today they are drawn in statistics programs as confidence regions (see below). The closer the data point for an object is to the center of a region, the more likely it is actually from that quarry. As more data has been taken, however, the field increasingly overlap, and stable isotope ratios alone are no longer considered conclusive evidence.
Today, most scientists employ multiple methods to determine chemical small differences between marble from different quarries, usually focusing on trace element analysis. This study is often referred to known as marble provenance. Other methods include [4]:
- Electron Paramagnetic Resonance (EPR) spectroscopy (also known as Electron Spin Resonance spectroscopy or ESR) - identifies unpaired electrons, which can show elemental content, such as the presence of magnesium to show whether marble is dolomitic. This method does not have a universal standard, however, so the data from different labs are not comparable, but efforts to standardize EPR data is underway. Today, the technique is less common as the instrument is large and costly (See image below).
- X-ray fluorescence (XRF) - identifies heavier elements. This method is primarily useful for identifying magnesium content to determine whether marble is dolomitic. Portable XRF machines have been developed, allowing for the instrument to be brought to museums for studying objects in situ.
- X-Ray diffraction (XRD) - identifies crystal structures. This method has limited applications, but like XRF, it can identify the mineral type.
- Trace element analysis - identifies elements besides calcium carbonate in marble. Similar to stable isotope analysis, it requires small samples and is relatively cheap. Trace elements are not always homogenously distributied within a quarry, or even within one sample of marble.
- Fluid inclusion chemistry - identifies the chemistry of microscopic fluids within marble. Unlike trace elements, fluid inclusions tend to be more homogeoneous, but it requires specialized laboratory instruments and large samples.
- Neutron activation analysis - determines the concentrations of elements in a sample using a nuclear reactor. While work has been done on ancient marble using reactors, it is increasingly difficult to get time on them for research, and the few studies using this method have not spurred further work.
- Cathodoluminescence - measures light emitted from a marble when exposed to an electron beam (cathode ray).
- Petrography - uses thin, transparent cross sections of marble to measure the physical structure, such as maximum grain size and whiteness magnitude.
[1] A valuable resource for mineralogy can be found at mindat.org, a mineralogy database containing information on the chemistry, 3D models of the crystal structures of calcite and dolomite, and more. See https://www.mindat.org/min-859.html for calcite and https://www.mindat.org/min-1304.html for dolomite.
[2] This general overview of marble chemistry and its unique features is not fully captured in any one source, but the proceedings of the Association for the Study of Marble and Other Stones in Antiquity (ASMOSIA) are the best resource for those comfortable with scientific and historical literature. A good starting point is the proceedings of the first conference: Norman Herz and Marc Waelkens, Classical Marble: Geochemistry, Technology, Trade, (NATO ASI Series. Series E, Applied Sciences; No. 153. Dordrecht; Boston: Kluwer Academic Publishers, 1988).
[3] See inter alia, Norman Herz and David B. Wenner, “Tracing the Origins of Marble” in Archaeology, vol. 34, No. 5 (September/October 1981), 14-21.
[4] Yannis Maniatis, "Scientific techniques and methodoliges for the provenance of white marbles" in Physics Methods in Archaeometry eds. M. Martini, M. Milazzo, and M. Piacentini (SIF Bologna – IOS Press, Amsterdam, 2004), 179-202.